What does it take to get the world vaccinated against COVID-19?
29.10.21
IP is part of the solution to get the world vaccinated against COVID-19: unprecedented R&D efforts and cooperation are making it possible for industry to produce over 12 billion vaccine doses by December 2021
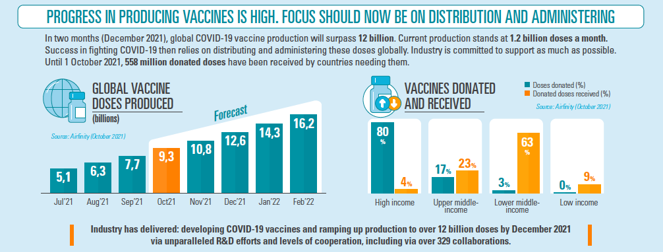
COVID-19 has shown the incredible complexity of the vaccine ecosystem, but the crisis has also driven a powerful response at all levels on R&D, production and vaccination. The vaccine industry has worked non-stop from day one of the pandemic to engage in R&D and then production to deliver safe and effective vaccines in record time around the world.Since the COVID-19 pandemic was declared, production has increased to 9.3 billion (October 2021), increasing by over 1.2 billion per month. At this rate, by December 2021 that figure will rise to over 12 billion doses. Exports are increasing rapidly, with EU-based producers having exported over 1 billion vaccines to date. Over 500 million doses have been donated to lower-middle and low-income countries so far and is rising rapidly.

These unprecedented feats could never have been achieved so quickly without a research-based pharmaceutical industry supported by a strong IP system, through voluntary and collaborative partnerships between the innovators and expert manufacturing partners behind each vaccine. IP has enabled the mRNA and adenovector technologies; allowed for approval of a first vaccine within 326 days; allowed the ramp-up of production we see today; facilitated technology transfers; reduced the risk of counterfeit vaccines; and supported global preparedness for new Variants of Concern of the virus. And yet, discussions on a COVID-19 waiver at the WTO still seem to continue despite having been fully overtaken by the facts.
Such calls, if approved by the WTO, would remove incentives for companies to continue research into new variants, new diagnostics, treatments and vaccines to tackle the coronavirus. At the same time, it would not only fail to increase the global capacity to manufacture COVID-19 vaccines, but risk current production levels because collaborations become more difficult and competition for limited resources between established producers and new start-up producers would intensify. In addition, removing patents on COVID-19 vaccines would also negate any innovation-based response to new variants and future pandemics.
A much better focus of the energy and discussions at the WTO than an obsolete TRIPS waiver is to discuss how to urgently advance COVID-19 vaccine equity with effective rather than symbolic measures. This can be a reality if all stakeholders work commit to the Five Steps to urgently advance COVID-19 vaccine equity which calls for stepping up dose sharing, eliminating trade barriers, and further supporting for country readiness. At the same time, industry is also working tirelessly to continue optimizing production and driving further innovation.