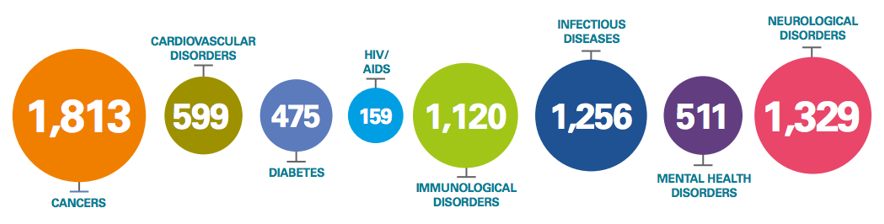
Value of medicines
Medicines and vaccines are some of the most powerful tools in helping people all over Europe live longer, healthier and more productive lives. Since the 1980’s we have seen death rates from HIV fall by over 80%, since the 1990’s deaths from cancer have fallen by 20% and recent pharmaceutical innovation means 95% of people living with Hepatitis C can be cured through an 8-12 week course of medicines.
These advances are helping us to address unmet need and tackle the world’s most debilitating diseases. Giving new hope and transforming the lives of patients.
And research is not stopping. The unprecedented speed of innovation provides an important opportunity for further improvement of outcomes for patients.
Through the Power of Innovation series, we are exploring the real impact of innovation; on patients, on healthcare systems and society as a whole.

Source: Health Advances analysis; [1] PhRMA 2016 Prescription Medicines: Costs in Context; [2] WHO Mortality Database (accessed February 2016). [2] EFPIA 2015 Health & Growth Evidence Compendia analysis of PhRMA 2014 25 Years of Progress Against Hepatitis C and PhRMA 2015 Pharma Profile