World IP Day 2026: keeping Europe on track for innovation
As Europe strives to compete globally in life sciences, predictable and robust IP incentives will determine whether tomorrow’s breakthroughs are developed here or elsewhere.
As Europe strives to compete globally in life sciences, predictable and robust IP incentives will determine whether tomorrow’s breakthroughs are developed here or elsewhere.
This year’s World IP day, with its theme “Ready, Set, Innovate.” is a timely reminder of Europe’s precarious position in life sciences.

Staying competitive in the global market and bringing new medicines to patients demand the right conditions to succeed and cannot be taken for granted. To do this it’s vital that we have an even playing field – the right policies and the right incentives to attract global companies to research. IP is a critical part of this.
The last Nobel Prize for economic sciences at the end of 2025 was awarded to researchers who deepened our understanding of how technological progress underpins innovation.
Unveiling the winners, the Nobel committee focused on how ‘new products and production methods replace others in a never-ending cycle’ – and celebrated the value of sustained growth in delivering better standards of living, health and quality of life.
But progress cannot be taken for granted. One of the stand-out lines in the announcement included this striking observation: ‘Stagnation was the norm throughout most of human history.’
It is tempting to be complacent about growth as it has become a feature of developed economies since the Industrial Revolution. We have lived through a time when, on average, living standards have steadily improved. Rather than presuming that this trend will continue indefinitely, we should consider the conditions that have delivered this remarkable achievement.
According to the Nobel Prize committee, the work of this year’s winners in economics points to the key role of creative destruction – the phenomenon through which new innovations and technologies replace old ones. This concept captures the creative side of inventing new things and ways of working, as well as the destruction that comes with making an established technology obsolete. The prize-winning work identified a key enabler of this productive cycle: effective incentives embedded in a functional and robust intellectual property system.

Pharmaceutical companies are familiar with both sides of this coin: when investments result in a breakthrough, they benefit; when a competitor develops a better alternative, they lose out. In some instances, a company or an entire industry can go out of business altogether if a truly transformative innovation comes to market. While the benefits for society and companies can be significant, the risks involved for companies are real.
Most of the chemical compounds discovered during the development process do not become medicines. In fact, around one in 10,000 make it into the development phase to potentially become a medicine. The likelihood that this compound passes all stages of pre-clinical and clinical testing is around 1%. As well as time, this costs developers between €2.4 billion and €3 billion. Ultimately, society only pays for successful medicines, while pharmaceutical companies must pay for all the failed ones.
That’s why incentives are needed. The biggest incentive for innovation is intellectual property protection such as patents (which offer market exclusivity for a limited time) and regulatory data protection (which protects research data being shared with follow-on competitors).
We should be clear about what market exclusivity means. If a company develops a new treatment for kidney cancer, they have a time-limited period during which competitors cannot copy and sell their product. But this does not prevent another company from making a new patentable innovation that might make earlier inventions obsolete. If a new, better product arrives on the market, the original patent can, and often does, become worthless.
Just as importantly, the IP system spreads knowledge. This is because the information required to be disclosed in order to be issued a patent can be freely used by the public after the IP rights have expired. That allows other companies to build on that science and develop the next generation of treatments.

Competition in the market is the great driver of progress, yet temporary protection from direct competition – through time-limited exclusivity – is essential to ensure future investment.
Getting the incentives right is a delicate balance, and intellectual property systems have sometimes been subject to criticism. EFPIA commissioned Sidley to explore the strengths and weaknesses of the current market-based system, and to look at how alternative frameworks could impact innovation-driven growth.
Innovation requires the capacity to incentivise large investments, at a significant financial risk, in the most efficient way possible. With over 100 medicines approved every year and more than 12,000 medicines currently in development, the EU IP framework is serving its objective in delivering new and improved treatments to patients who need them.
The impact of this innovation has been enormous: life expectancy in Europe has increased by nearly a decade over the last 50 years; two out of three people diagnosed with cancer live at least five years; HIV has been transformed from a death sentence to a manageable disease; and over 90% of Hepatitis C sufferers can be cured with a 12-week course of medicine. Even remarkably high-risk areas such as Alzheimer’s Disease, where more than 99% of clinical trials fail, continue to attract investment.
We are in an era of exciting, ground-breaking science that has the potential to open up new treatment options for patients in disease areas previously thought to be untreatable. None of these significant, life-changing breakthroughs could have been achieved without the market-based IP framework that exists in the EU today.

As we speak, Europe is rewriting the rules that will define the future of our sector. At a time when companies are already navigating a complex maze of regulatory reforms, cost-containment policies, environmental obligations, tariff risks, and geopolitical uncertainty, legislation must relieve pressure, not add to it. Weakening intellectual property protections risk making it harder to invest, harder to innovate, and ultimately harder to bring new medicines to European patients.
Against this backdrop, Sidley’s analysis shows that while alternatives may exist in theory, none can match the effectiveness, efficiency or reliability of today’s market-based, IP-driven system. One alternative proposed by critics of the current system is to nationalise pharmaceutical research and production. However, this demands technical and financial capacity that public institutions do not have. Public infrastructure is also broadly viewed by economists as less efficient in allocating investment compared to market-based approaches.
Replacing the private sector with a government-funded model has another obvious downside: it puts public funding at risk and is financed either by government debt or higher tax revenue.
So, while public and private organisations collaborate productively on pre-clinical research, helping to raise standards in industry and academia, the wholesale replacement of private sector innovation is an inefficient alternative. The risk is not only that governments would face financial strain, but that the flow of innovation would dry up – with widespread health and economic consequences.
Innovation prizes have also been suggested as a possible alternative. Such prizes are generally awarded for ideas or prototypes. In practice, they suffer from multiple drawbacks: it is extremely difficult to calculate the value of a new medicine in advance; prizes necessarily imply significant duplication of effort; and the question of how to fund them remains unresolved.
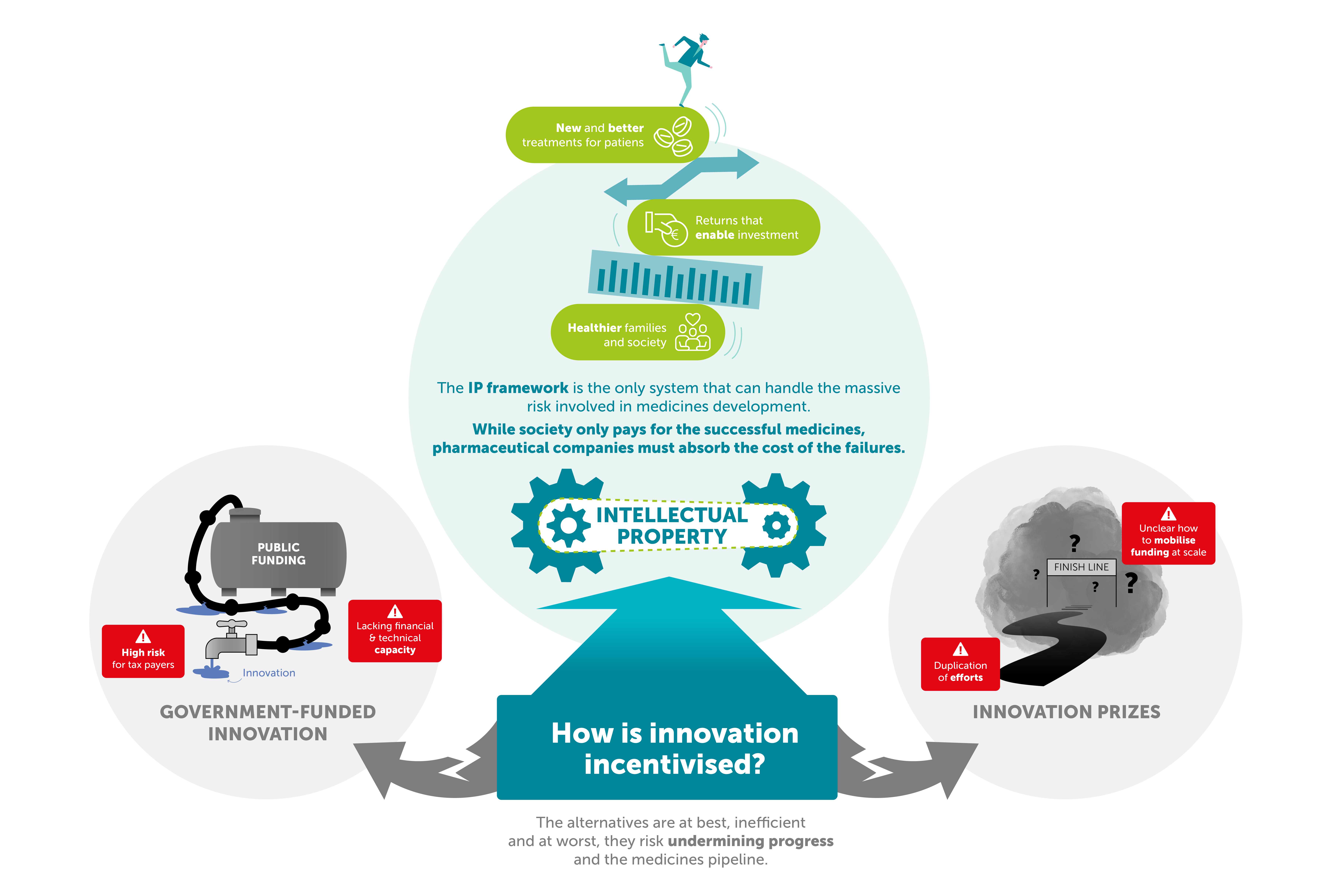

The Nobel Prize is a timely reminder of the value of innovation and the vital role played by our current incentives system, with intellectual property and disclosure-protection at its heart. Market-based innovation, based on patents, is the best mechanism we have for driving medical advances. The alternatives are, at best, inefficient. At worst, they risk undermining progress.
As Europe strives to compete globally in life sciences, predictable and robust IP incentives will determine whether tomorrow’s breakthroughs are developed here or elsewhere. Legislative initiatives such as the Biotech Act provide an opportunity to create new incentives that will help Europe realise its ambitions to be a world leader in life sciences.
If you would like to go more in depth on the underlying work behind this study, please reach out to Michael Swita at michael.swita@efpia.eu.

