EFPIA update on research-based pharmaceutical industry response to COVID-19
11.03.20
As the EU leaders hold emergency talks to coordinate a Europe-wide response to the coronavirus epidemic, thirteen leading biopharmaceutical companies have announced their participation in the Innovative Medicines Initiative (IMI) coordinated efforts to find new treatments and diagnostics. IMI, the world’s largest public-private partnership in health, launched the fast-track call 21 in early March investing €45 millions of EU funds, which will be complemented by investment from participating companies.
The call 21 will bring together companies, academia, international organisations and public bodies to accelerate the development of therapeutics and diagnostics to tackle the current and future outbreaks of coronavirus.
Since a pneumonia of unknown cause was detected in Wuhan, China and reported to the WHO Country Office in China on 31 December 2019, there have been 113,702 confirmed cases and 4,012 deaths from coronavirus[1]. The outbreak was declared a Public Health Emergency of International Concern on 30 January 2020. On 11 February 2020, WHO announced a name for the new coronavirus disease: COVID-19. Although originating in China, the latest update from WHO shows the truly global nature of this outbreak as well as its prominence in Europe.
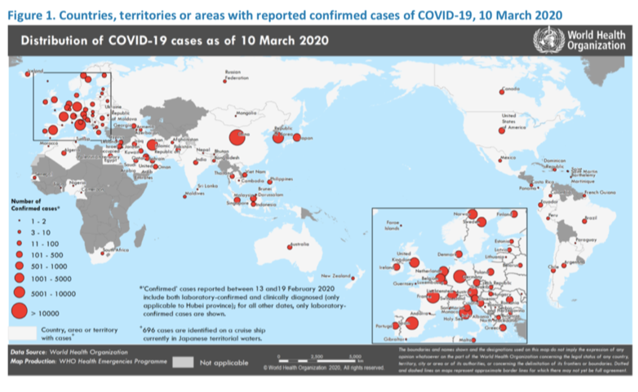
The global research and development effort mobilizing to fight COVID-19 can utilize and build on previous research and infrastructure. Experience in fighting SARS, MERS, and Ebola have paved the way for collaboration to accelerate development of resources to tackle this new outbreak. For example, the IMI response to the Ebola crisis in West Africa pioneered a collaborative approach that resulted in new diagnostic tests, vaccines and future outbreak preparedness strategies.
The IMI projects will contribute to the pan-European efforts responding to this Public Health Emergency by developing solutions that can be deployed in a very short term but also preparing for future outbreaks. This may include antiviral agents as well as symptom relief medicines and rapid point of care and lab based diagnostics for detection of carriers and symptomatic individuals.
The companies already engaged in IMI Call 21 on COVID-19 include Abbvie, Astellas, Bayer, Boehringer Ingelheim, E-Pharma, Enyo Pharma, IDbyDNA, Janssen, Merck, Novartis, Pfizer, Special Product's Line S.p.A. Dompe’ S.p.A. and Takeda. Outreach continues to additional partners – pharmaceutical and diagnostic companies and other investors.
This unprecedented collaborative effort with so many companies joining forces in a precompetitive context includes companies that are not traditionally involved in the infectious disease space and want to share molecules that might have an anti-COVID effect. This complements the rapid development programmes that could be very quickly mobilised by vaccines, biotech and pharmaceutical companies – as a result several vaccines and medicines could be moved to final development phases in dialogue with global funders, governments and regulators.
In addition to the research and development efforts, EFPIA member companies are collaborating with relevant European and global public health authorities including the European Medicines Agency, National Health Authorities across Europe as well as the World Health Organization (WHO), Centres for Disease Control and Prevention (CDC), Chinese public health authorities including the Chinese Centre for Disease Control and Prevention and many others to address this public health crisis.
EFPIA member companies continue to donate millions of euros in direct monetary and in-kind contributions to support organizations at the heart of the crisis who are able to have an immediate impact for infected patients and communities. EFPIA member companies acted immediately on the ground to donate a variety of crucial supplies including advanced surgical equipment, antibiotics, disinfection equipment, batch virus testing devices (e.g., throat swabs), vitamins, protective clothing, goggles, masks, gloves and more.
The advent of a global health emergency such as coronavirus underlines the need for a collaborative, dynamic, responsive and empowered research eco-system in Europe. As a key part of that eco-system, the research-based pharmaceutical industry will continue to strive to develop a vaccine, diagnostics and treatments for coronavirus.
[1] WHO, Coronavirus disease 2019 (COVID-19), Situation Report – 50, https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200310-sitrep-50-covid-19.pdf?sfvrsn=55e904fb_2